Drug Information
| General Information | ||||||
|---|---|---|---|---|---|---|
| Drug ID |
DR01281
|
|||||
| Drug Name |
Rizatriptan
|
|||||
| Synonyms |
MK 462 free base; Maxalt (TN); N,N-Dimethyl-2-[5-(1H-1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]ethylamine; N,N-Dimethyl-5-(1H-1,2,4-triazol-1-ylmethyl)-1H-indole-3-ethanamine; N,N-dimethyl-2-[5-(1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]-ethanamine; N,N-dimethyl-2-[5-(1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]ethanamine; N,N-dimethyl-2-[5-(1H-1,2,4-triazol-1-ylmethyl)-1H-indol-3-yl]ethanamine; Risatriptan; Rizaliv (TN); Rizalt (TN); Rizatriptan (INN); Rizatriptan [INN:BAN]; Rizatriptan benzoat; Rizatriptanum
|
|||||
| Drug Type |
Small molecular drug
|
|||||
| Indication | Migraine Headaches [ICD11:8A80] | Approved | [1] | |||
| Therapeutic Class |
Antiinflammatory Agents
|
|||||
| Structure |
|
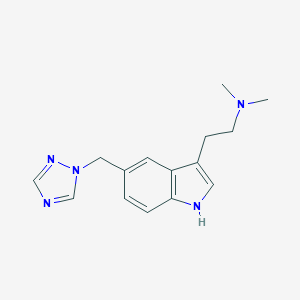 |
||||
| 3D MOL | 2D MOL | |||||
| Formula |
C15H19N5
|
|||||
| Canonical SMILES |
CN(C)CCC1=CNC2=C1C=C(C=C2)CN3C=NC=N3
|
|||||
| InChI |
InChI=1S/C15H19N5/c1-19(2)6-5-13-8-17-15-4-3-12(7-14(13)15)9-20-11-16-10-18-20/h3-4,7-8,10-11,17H,5-6,9H2,1-2H3
|
|||||
| InChIKey |
ULFRLSNUDGIQQP-UHFFFAOYSA-N
|
|||||
| CAS Number |
CAS 145202-66-0
|
|||||
| Pharmaceutical Properties | Molecular Weight | 269.34 | Topological Polar Surface Area | 49.7 | ||
| Heavy Atom Count | 20 | Rotatable Bond Count | 5 | |||
| Hydrogen Bond Donor Count | 1 | Hydrogen Bond Acceptor Count | 3 | |||
| XLogP |
1.7
|
|||||
| PubChem CID | ||||||
| PubChem SID |
7380056
,7980527
,8153125
,15221855
,26612819
,26749916
,29224147
,46506557
,46530620
,48416524
,49830967
,49984083
,50173225
,57322597
,85209524
,92718843
,93166519
,96025171
,99373553
,103236413
,103941727
,104308214
,125001917
,125728524
,126621280
,126658125
,126667003
,129384422
,131299041
,134337535
,134358454
,135110600
,135650905
,135684127
,136375513
,137002445
,139157639
,143494842
,144205087
,152036015
,152239970
,160964292
,162011662
,172091432
,174007052
,174477513
,174527522
,175266240
,176484738
,179116883
|
|||||
| ChEBI ID |
ChEBI:48273
|
|||||
| TTD Drug ID | ||||||
| DT(s) Transporting This Drug | 1-Oct | Transporter Info | Organic cation transporter 1 | Substrate | [2] | |
| References | ||||||
| 1 | Rizatriptan was approved by FDA. The official website of the U.S. Food and Drug Administration. (2019) | |||||
| 2 | OCT1 mediates hepatic uptake of sumatriptan and loss-of-function OCT1 polymorphisms affect sumatriptan pharmacokinetics. Clin Pharmacol Ther. 2016 Jun;99(6):633-41. | |||||
If you find any error in data or bug in web service, please kindly report it to Dr. Li and Dr. Fu.
